Galvanized Coatings & Corrosion Resistance for Woven Fence Wire
Galvanized coatings represent the primary corrosion defense mechanism for steel fence wire installations across agricultural, ranch, and perimeter security applications. The performance durability of woven wire fence systems depends fundamentally on zinc coating integrity, environmental exposure conditions, and maintenance protocols. This technical resource examines the electrochemical protection mechanisms, coating classification standards, service life projections, and practical selection criteria for specifying galvanized fence wire in diverse operational environments.
What Is Galvanization and Why It Matters
Galvanization constitutes a metallurgical coating process that deposits zinc onto steel surfaces through immersion in molten zinc baths (hot-dip galvanizing) or electrochemical deposition (electroplating). The zinc coating serves dual protective functions: it establishes a physical barrier preventing atmospheric moisture and oxygen from contacting the underlying steel substrate, and it provides electrochemical sacrificial protection when coating damage exposes the base metal.
Hot-Dip Galvanization Process
Hot-dip galvanization involves immersing cleaned steel wire into molten zinc maintained at approximately 450°C (840°F). During immersion, the iron in steel reacts with molten zinc to form a series of zinc-iron alloy layers metallurgically bonded to the substrate. This reaction creates coating thicknesses ranging from 50 to 150 micrometers depending on steel composition, immersion duration, and zinc bath chemistry. The resulting coating exhibits superior adhesion and abrasion resistance compared to applied coatings, with bond strengths exceeding 3,600 psi.
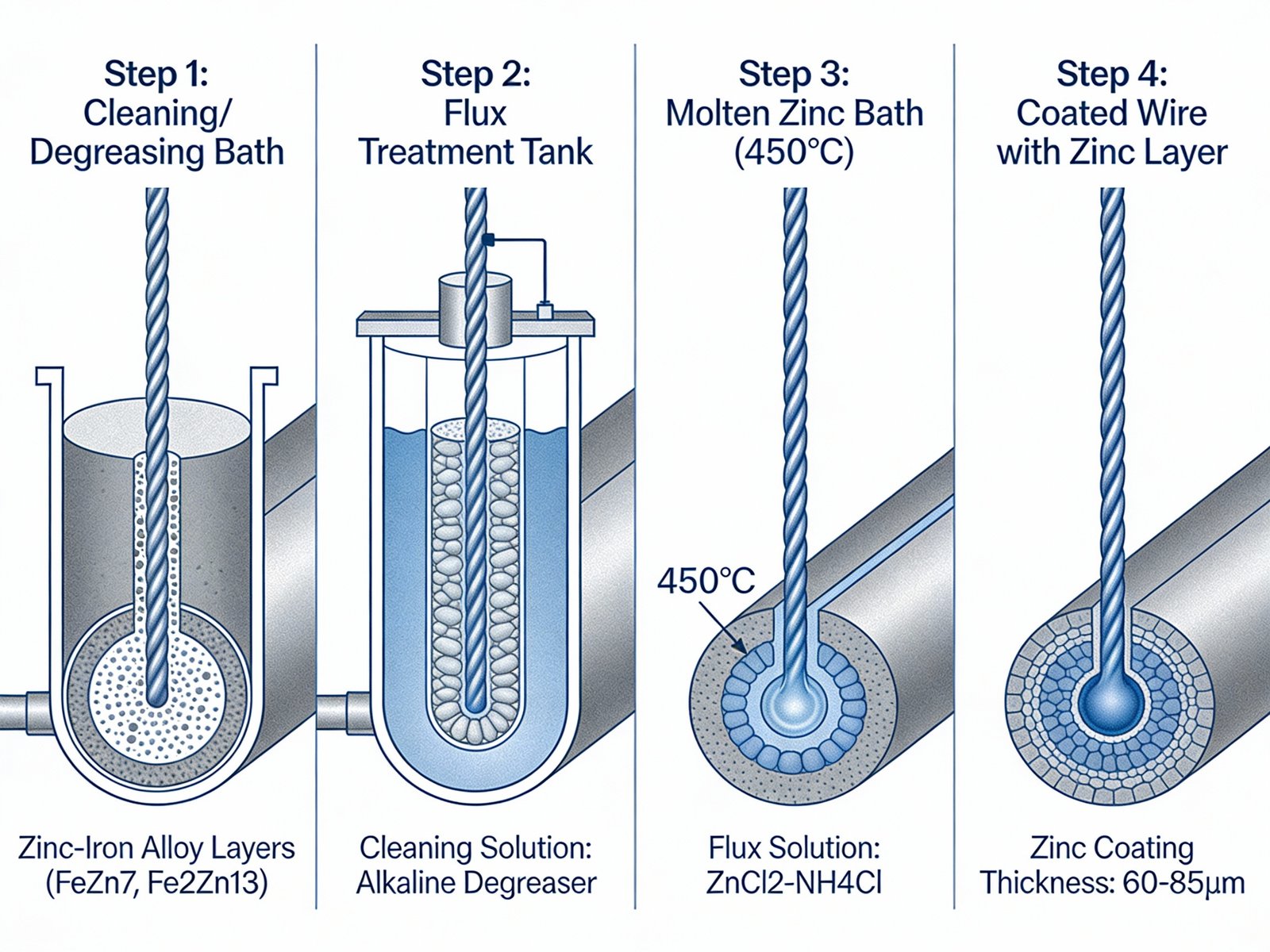
Figure 1: Technical diagram illustrating the hot-dip galvanization vs electroplating process steps for fence wire, showing steel immersion in molten zinc bath, zinc coating formation, and galvanic protection mechanism
Electroplating Alternative
Electroplating deposits zinc through electrochemical reduction in an aqueous zinc salt solution. While this method produces uniform, thin coatings suitable for aesthetic applications, the coating thickness typically measures only 0.0003 inches (7.6 micrometers) compared to hot-dip galvanizing’s minimum 0.002 inches (50 micrometers). Electroplated zinc coatings provide significantly reduced corrosion protection, with expected service lives of 2 to 11 years versus 20 to 75+ years for hot-dip galvanized steel in equivalent environments. The mechanical bond strength of electroplated coatings averages only 400-600 psi, making them vulnerable to damage during installation and livestock contact.
The Sacrificial Protection Principle
Zinc occupies a more anodic (electronegative) position than iron in the galvanic series. When both metals contact an electrolyte—such as moisture condensation, rainfall, or humid air—zinc preferentially corrodes while the steel cathode remains protected. This galvanic protection extends several millimeters beyond the point of coating damage, preventing the rust “creep” characteristic of organic coatings. In normal atmospheric conditions, zinc corrodes at approximately one-tenth the rate of unprotected carbon steel, providing proportional service life extension relative to coating thickness.
How Galvanized Coatings Protect Steel Fence Wire
Electrochemical Corrosion Fundamentals
Steel corrosion proceeds through electrochemical oxidation-reduction reactions requiring four elements: an anode (corroding metal), a cathode (protected metal), an electrolyte (conductive solution), and an electrical connection. In unprotected steel, microscopic variations in surface composition create local anodic and cathodic sites. The anodic reaction oxidizes iron atoms to ferrous ions (Fe → Fe²⁺ + 2e⁻), releasing electrons that migrate to cathodic sites where they reduce oxygen in the presence of water (O₂ + 2H₂O + 4e⁻ → 4OH⁻). The ferrous ions combine with hydroxide ions to form iron hydroxide, which oxidizes further to ferric oxide—commonly recognized as rust.
Barrier Protection Mechanism
The zinc coating’s primary protective function derives from its impermeability to oxygen and water vapor. The dense metallic structure prevents atmospheric moisture from reaching the steel surface, eliminating the electrolyte required for corrosion. Upon atmospheric exposure, the pure zinc surface rapidly oxidizes to zinc oxide, which subsequently reacts with atmospheric moisture and carbon dioxide to form zinc hydroxide and zinc carbonate. These corrosion products possess very low solubility in water and form a tightly adherent passive layer—the zinc patina—that substantially reduces further zinc corrosion rates.
Sacrificial Corrosion and Cathodic Protection
When mechanical damage, abrasion, or wire cutting exposes the steel substrate, the adjacent zinc coating establishes a galvanic cell with the exposed steel. The zinc functions as a sacrificial anode, corroding preferentially while maintaining the steel in a cathodic (protected) state. This protection remains effective as long as zinc coating exists within several millimeters of the exposed area. Field observations demonstrate that scratches, cut ends, and damaged areas on galvanized fence wire do not develop progressive rust penetration, contrasting sharply with painted or polymer-coated wires where coating breaches initiate rapid localized corrosion.
Coating Thickness and Corrosion Delay
Zinc corrosion in atmospheric environments proceeds at a relatively constant linear rate for a given location and exposure condition. Consequently, coating service life demonstrates direct proportionality to zinc coating thickness. Empirical data from long-term atmospheric exposure studies indicate that doubling the zinc coating mass yields approximately double the time to coating depletion. This linear relationship enables reliable service life prediction when environmental corrosivity is characterized, providing the engineering basis for coating class selection in fence wire specifications.
ASTM Zinc Coating Classes & Durability Expectations
The American Society for Testing and Materials (ASTM) establishes standardized coating weight classifications that govern fence wire galvanization specifications. These standards—primarily ASTM A121 (barbed wire), A641 (carbon steel wire), and A817 (chain link fabric wire)—define minimum zinc coating weights expressed in ounces per square foot of uncoated wire surface area. Understanding these classifications proves essential for lifecycle cost analysis and performance specification.
Class 1 Coating Specification
Class 1 galvanized wire carries a minimum zinc coating weight of 0.28 ounces per square foot (85 grams per square meter) of uncoated wire surface. This represents the minimum acceptable coating for outdoor fence applications. Class 1 coating typically produces a zinc layer approximately 20-25 micrometers thick. In non-coastal temperate climates with moderate humidity, Class 1 galvanized fence wire exhibits visible rust formation within 4 to 12 years depending on local conditions. Coastal installations experience significantly accelerated deterioration, with rust onset occurring within 2 to 3 years. Class 1 coating finds appropriate application in temporary fencing, budget-constrained projects with short design life requirements, or installations in arid low-corrosivity environments.
Class 3 Coating Specification (Industry Standard)
Class 3 galvanized wire specifies a minimum zinc coating weight of 0.80 ounces per square foot (244 grams per square meter)—approximately 2.86 times the zinc mass of Class 1 coating. This heavier coating produces zinc layers 55-65 micrometers thick, providing substantially extended corrosion resistance. Class 3 represents the prevailing industry standard for permanent agricultural and ranch fencing, offering projected service lives of 13 to 30 years in non-coastal environments with typical atmospheric corrosivity. Coastal applications of Class 3 wire demonstrate service lives of 8 to 15 years depending on proximity to salt exposure. The incremental material cost of Class 3 versus Class 1 wire—typically 15-25% higher initial purchase price—yields significant lifecycle cost advantages through extended replacement intervals and reduced maintenance.
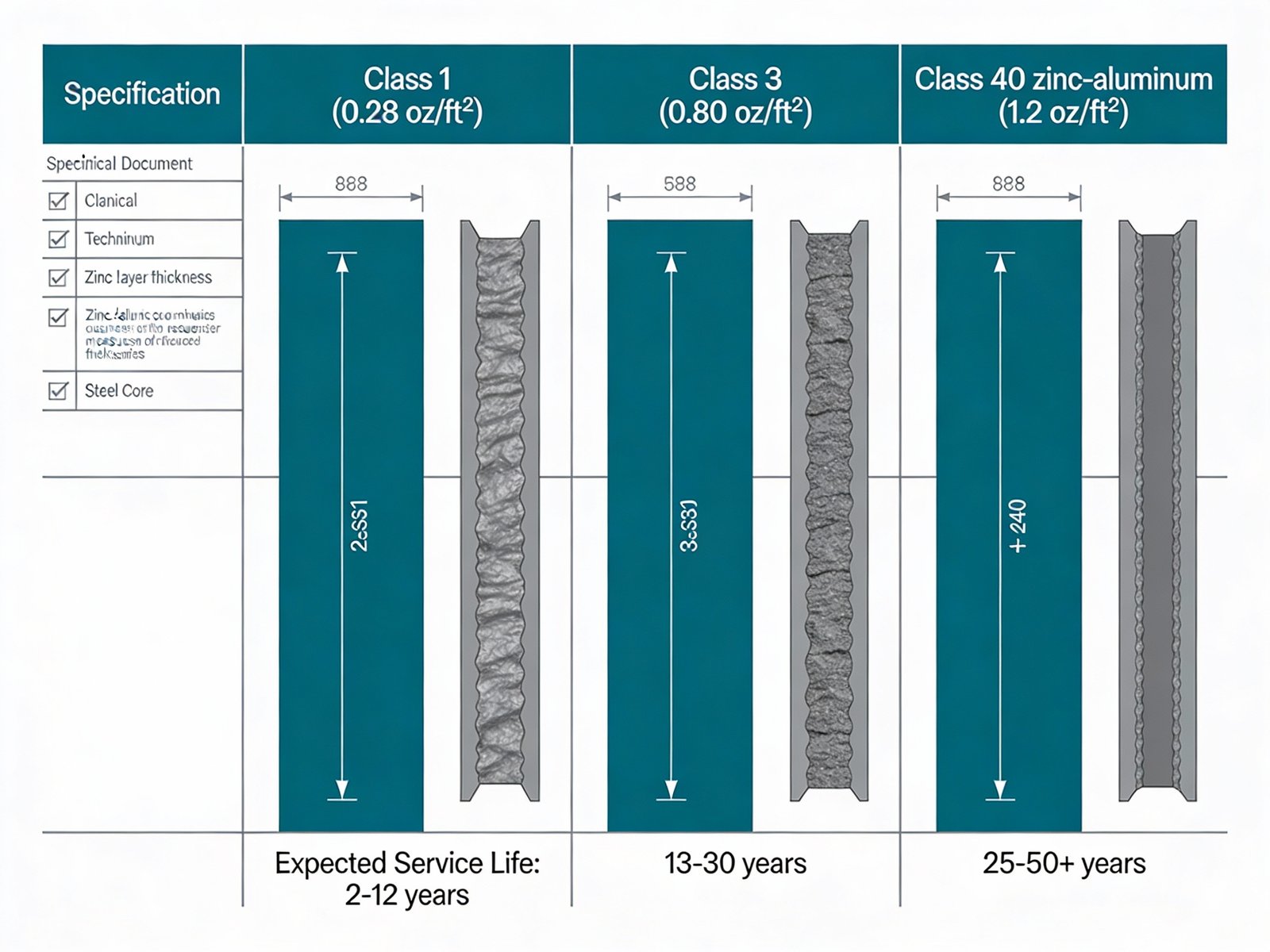
Figure 2: Data visualization comparing zinc coating weight per square foot for Class 1, Class 3, and Class 40 zinc-aluminum coatings and corresponding expected service life ranges
Class 40 Zinc-Aluminum Coating
Class 40 coating employs a zinc-aluminum alloy composition consisting of approximately 95% zinc and 5% aluminum by weight. The coating weight typically reaches 1.2 to 1.4 ounces per square foot, with the aluminum component enhancing both barrier protection and corrosion product formation. Aluminum additions modify the zinc patina chemistry, producing corrosion products with superior adhesion and protective characteristics. Class 40 zinc-aluminum coatings provide service life projections of 25 to 50+ years in moderate environments, with coastal installations achieving 15 to 30 years before requiring replacement. The coating exhibits a characteristic matte gray appearance rather than the bright metallic sheen of pure zinc coatings. While initial material costs exceed Class 3 wire by 40-60%, applications in highly corrosive environments or installations requiring maximum design life justify the premium through extended replacement cycles.
| Coating Class | Zinc Weight (oz/ft²) | Typical Thickness (μm) | Service Life: Inland | Service Life: Coastal | ASTM Standards |
|---|---|---|---|---|---|
| Class 1 | 0.28 | 20-25 | 4-12 years | 2-3 years | A121, A641, A817 |
| Class 3 | 0.80 | 55-65 | 13-30 years | 8-15 years | A121, A641, A817 |
| Class 40 (ZnAl) | 1.2-1.4 | 85-100 | 25-50+ years | 15-30 years | Proprietary/Enhanced |
Service Life Variables and Field Performance
Published service life ranges reflect substantial variability because actual coating performance depends on site-specific environmental factors. Atmospheric corrosivity classifications defined in ISO 9223 characterize environments from C1 (very low corrosivity, arid interiors) through C5 (very high corrosivity, coastal/industrial with persistent wetness). Zinc corrosion rates vary by factors of 10 to 20 across this spectrum. Consequently, a Class 3 galvanized fence wire installed in a C2 (low corrosivity) rural environment may achieve 30+ years of service, while an identical wire in a C4 (high corrosivity) coastal location may exhibit coating failure within 10 years. Proper coating class selection requires environmental assessment of the specific installation site rather than reliance on generic lifespan claims.
Environmental Factors Influencing Corrosion
Galvanized coating degradation rates depend critically on atmospheric and soil chemistry, moisture availability, temperature cycling, and pollutant exposure. Quantifying these environmental stressors enables evidence-based coating selection and realistic service life projection for fence installations.

Figure 3: Environmental exposure map illustrating how coastal salt air, high humidity inland, and arid ranch conditions differentially impact steel fencing corrosion rates
Coastal and Marine Environments
Coastal installations face the most aggressive corrosion conditions encountered in fence applications. Airborne sodium chloride particles from ocean spray travel miles inland, settling on fence surfaces where they attract atmospheric moisture to form highly conductive electrolyte solutions. Chloride ions penetrate zinc oxide films and accelerate zinc dissolution through electrochemical mechanisms. Properties within one-quarter mile of coastlines experience severe marine exposure, with zinc corrosion rates 5 to 10 times higher than inland locations. Coastal effects extend 1 to 3 miles inland depending on prevailing wind patterns and topography, with salt deposition declining exponentially with distance. Standard Class 1 galvanized wire proves inadequate for coastal applications, typically failing within 2 to 3 years. Class 3 coating extends service life to 8 to 15 years, while Class 40 zinc-aluminum provides optimal performance for marine environments. Additional protective measures—including polymer topcoats over galvanizing—merit consideration for installations within direct salt spray zones.
Humidity and Precipitation Patterns
Atmospheric moisture governs zinc corrosion kinetics by providing the electrolyte necessary for electrochemical reactions. Regions with relative humidity consistently above 60% maintain nearly continuous surface wetness through dew formation and hygroscopic salt deposits, sustaining active corrosion. Conversely, arid climates with low humidity and infrequent precipitation dramatically reduce zinc corrosion rates, with fence wire in desert environments often exceeding service life projections by factors of two to three. Acid precipitation containing dissolved sulfur dioxide and nitrogen oxides increases zinc corrosion by lowering the pH of surface moisture. Industrial and urban environments with elevated atmospheric pollutants typically exhibit 2 to 3 times higher zinc corrosion rates compared to rural areas with equivalent precipitation and humidity.
Soil Chemistry and Ground Contact
Fence wire sections in direct soil contact experience localized corrosion acceleration dependent on soil chemistry. Soil pH represents the dominant variable, with zinc demonstrating maximum corrosion resistance in the pH range of 5.5 to 12.5. Acidic soils below pH 5.5 dissolve the protective zinc carbonate patina, exposing bare zinc to continuous attack. Alkaline soils above pH 12.5 similarly compromise zinc stability, though such extreme alkalinity occurs rarely in natural agricultural soils. Soil moisture content governs corrosion rate by controlling electrolyte availability; waterlogged soils promote rapid zinc dissolution while dry soils provide minimal corrosive activity. Soil electrical resistivity—influenced by moisture content and dissolved salt concentration—correlates inversely with corrosion rate. Soils with resistivity below 1,000 ohm-cm exhibit severe corrosivity, while resistivity above 10,000 ohm-cm indicates minimal corrosive potential. Fence installations incorporating wire spacing & tension design principals that minimize ground contact reduce bottom wire corrosion substantially.
Temperature Cycling and Seasonal Effects
Diurnal and seasonal temperature fluctuations induce expansion and contraction stresses in zinc coatings while cycling moisture condensation on wire surfaces. Freeze-thaw cycling proves particularly detrimental, as ice crystal formation within coating microcracks propagates damage. Regions experiencing frequent freeze-thaw transitions—typically 20 to 40 cycles annually in temperate climates—demonstrate accelerated coating degradation compared to locations with stable temperatures. Summer heat combined with high humidity maximizes zinc corrosion rates, while cold dry winter conditions suppress corrosive activity. Consequently, fence installations in humid continental climates with hot summers and cold winters face dual corrosion stresses that reduce service life projections compared to stable maritime or desert climates.
| Environment Type | Primary Corrosion Drivers | Relative Corrosion Rate | Class 3 Expected Life |
|---|---|---|---|
| Arid Inland (Desert/Ranch) | Low humidity, minimal precipitation, high UV | 0.5-1.0 μm/year | 25-30+ years |
| Temperate Inland (Rural) | Moderate humidity, seasonal precipitation | 1.0-2.0 μm/year | 18-25 years |
| Humid Inland (Agricultural) | High humidity, frequent precipitation, soil contact | 2.0-3.5 μm/year | 13-18 years |
| Coastal (< 1 mile from ocean) | Salt air, high humidity, chloride exposure | 4.0-8.0 μm/year | 8-12 years |
| Industrial/Urban | Acid rain, SO₂/NOₓ pollution, high humidity | 3.0-5.0 μm/year | 10-15 years |
Comparing Corrosion Resistance Across Coating Types
Hot-Dip Galvanizing Performance Profile
Hot-dip galvanizing establishes the performance standard for fence wire corrosion protection due to its thick metallurgically bonded coating, comprehensive coverage including wire intersections and weld points, and superior mechanical durability. The coating’s zinc-iron alloy layers exhibit hardness exceeding the base steel substrate, providing excellent abrasion resistance during installation, livestock contact, and vegetation management operations. Hot-dip galvanized wire accommodates field bending, cutting, and tie-off operations without coating spalling or delamination. The immersion process ensures uniform coating on complex geometries, including the critical fixed knot construction and hinge joint knot design intersections where crevice corrosion otherwise initiates. Cost-effectiveness analysis demonstrates that hot-dip galvanized Class 3 wire provides optimal lifecycle economics for permanent fence installations, balancing initial material cost against multi-decade service life.
Electroplated Zinc Limitations
Electroplated (electro-galvanized) fence wire carries significantly thinner zinc deposits, typically 5 to 10 micrometers versus 55+ micrometers for hot-dip Class 3 wire. This reduced thickness provides only 10-20% of the corrosion protection duration. Electroplated coatings demonstrate higher susceptibility to mechanical damage, with installation handling and post-installation livestock contact frequently abrading through the thin zinc layer to expose steel substrate. The electrochemical bond strength averages 400-600 psi compared to 3,600 psi for hot-dip coatings, resulting in adhesion failures under field stress. Electroplating proves economically rational only for indoor applications, temporary installations with design lives under 5 years, or aesthetic applications where appearance supersedes corrosion protection. Specifying electroplated wire for permanent outdoor fence construction constitutes a false economy, as premature replacement costs exceed the initial savings within the first decade of service.
Zinc-Aluminum Alloy Enhancements
Zinc-aluminum (ZnAl) coatings—designated Class 40—incorporate 5% aluminum into the zinc matrix, modifying both coating microstructure and corrosion product chemistry. The aluminum component forms aluminum oxide within the coating, which exhibits superior chemical stability compared to pure zinc oxide. Field exposure studies demonstrate that ZnAl coatings develop more adherent, protective patina layers that reduce the long-term zinc corrosion rate by 30-50% compared to pure zinc coatings of equivalent thickness. The aluminum addition also improves coating ductility, reducing microcracking during wire forming operations. ZnAl coatings exhibit particular advantage in coastal and industrial environments where chloride and sulfur dioxide exposures challenge pure zinc coatings. The premium cost—typically 40-60% above Class 3 galvanized wire—finds economic justification in high-corrosivity installations, critical infrastructure applications requiring maximum design life, and projects where replacement logistics impose high costs.
Polymer-Coated Galvanized Wire
Polymer-coated wires employ a PVC or polyethylene outer layer over a galvanized steel core, providing a composite protection system. The polymer barrier excludes moisture and oxygen when intact, while the underlying zinc coating provides backup protection if the polymer layer sustains damage. This dual-layer approach extends service life in extreme environments but introduces additional considerations. Polymer coatings exhibit sensitivity to UV radiation, requiring stabilizer additives to prevent photodegradation and embrittlement. Physical damage from vegetation management equipment, livestock horns, or installation handling can breach the polymer layer, potentially creating crevice corrosion sites between polymer and zinc if moisture infiltrates. Polymer-coated galvanized wire finds appropriate application in coastal installations, highly visible boundary fencing where aesthetics matter, and installations requiring color-coding. The wire costs 2 to 3 times more than bare galvanized Class 3 wire, requiring clear service life or aesthetic justification.
| Coating Type | Typical Thickness | Service Life Range | Relative Cost | Optimal Applications |
|---|---|---|---|---|
| Electroplated Zinc | 5-10 μm | 2-8 years | 1.0× | Indoor, temporary, budget installations |
| Hot-Dip Class 1 | 20-25 μm | 4-12 years | 1.2× | Arid environments, short-term projects |
| Hot-Dip Class 3 | 55-65 μm | 13-30 years | 1.4× | Standard permanent fencing (industry baseline) |
| Class 40 ZnAl | 85-100 μm | 25-50+ years | 2.0× | Coastal, maximum design life requirements |
| Polymer + Galvanized | 200-400 μm total | 20-40 years | 3.0-4.0× | Coastal, aesthetic requirements, high-value |
Practical Selection Guidance for Ranch & Farm Applications
Environmental Assessment Methodology
Evidence-based coating selection begins with systematic environmental characterization of the installation site. Evaluate distance from coastline (< 0.25 mile = severe exposure; 0.25-1 mile = high exposure; 1-3 miles = moderate coastal influence; > 3 miles = inland). Assess typical relative humidity during growing season (< 50% = low; 50-70% = moderate; > 70% = high corrosion risk). Characterize soil conditions for ground-contact wires through pH measurement and visual moisture assessment. Consider vegetation management practices that may cause mechanical coating damage. Document proximity to industrial facilities, major roadways with winter salt application, or other pollution sources. This environmental profile maps directly to appropriate coating class selection following the decision framework below.
Coating Class Selection Framework
Class 1: Limit application to temporary installations (< 5 year design life), budget-constrained projects accepting early replacement, or desert/arid environments with documented low corrosivity (annual precipitation < 10 inches, relative humidity consistently < 40%). Class 1 proves inadequate for permanent livestock or perimeter fencing in moderate to high corrosivity environments.
Class 3 (Recommended Standard): Specify for all permanent fence installations in temperate inland, rural agricultural, and moderate-humidity environments located beyond 3 miles from coastline. Class 3 provides optimal lifecycle economics for typical ranch and farm applications, delivering 15-25 year service life at modest cost premium over Class 1. Ensure specifications reference ASTM standards (A121, A641, or A817) with explicit Class 3 coating weight requirement.
Class 40 ZnAl: Specify for coastal installations within 3 miles of ocean, industrial/urban environments with documented air quality issues, properties with acidic soils (pH < 5.5), high-value applications justifying maximum design life, or installations where replacement logistics impose prohibitive costs. The material premium finds economic justification when lifecycle analysis accounts for replacement costs, fence downtime, and livestock management disruption.
Wire Grade and Coating Integration
Coating class selection must integrate with base wire specifications for tensile strength, carbon content, and knot configuration. High-tensile wire designs common in S knot structure fence require Class 3 minimum coating to prevent stress corrosion cracking at high-load points. Low-carbon wire grades accommodate heavier Class 40 coatings without embrittlement concerns. Review complete woven wire fence specifications to ensure coating and wire grade compatibility. Installations incorporating difficult terrain should reference installation in difficult terrain guidance to account for coating damage risks during construction.
Lifecycle Cost Analysis
Compare coating alternatives using total cost of ownership rather than initial purchase price. Calculate material cost per linear foot for each coating class. Project service life based on environmental assessment. Estimate replacement costs including materials, labor, fence removal/disposal, and livestock management disruption during replacement. Discount future costs using appropriate discount rate (typically 3-5% for agricultural capital investments). Analysis consistently demonstrates that Class 3 coating provides minimum lifecycle cost for permanent installations, with Class 40 proving economical in high-corrosivity environments where Class 3 service life falls below 15 years.
Maintenance Strategies to Maximize Service Life
Proactive maintenance extends galvanized fence wire service life by 30-60% compared to neglected installations. Systematic inspection and targeted intervention preserve coating integrity and delay replacement requirements.
Inspection Protocol and Schedule
Conduct visual fence inspections biannually—early spring following winter moisture exposure and late fall after vegetation management operations. Focus inspection attention on high-risk zones: bottom wires with soil contact, wire intersections at knots and splices, areas with dense vegetation contact, and sections within 100 feet of livestock watering points (elevated moisture). Document coating condition through photographic records, noting the progression of zinc patina formation (normal), white rust appearance (zinc corrosion products, concerning), or red rust breakthrough (steel exposure, critical). Properties in coastal or high-corrosivity environments warrant quarterly inspection intervals to enable early intervention before steel exposure occurs.
Early Corrosion Detection
Distinguish between normal zinc patina development and problematic corrosion progression. Freshly galvanized wire exhibits bright metallic zinc appearance that weathers to dull gray zinc patina within 6-18 months of exposure—this represents normal protective layer formation and does not indicate coating failure. White rust (zinc oxide/hydroxide) appears as chalky white deposits, indicating active zinc corrosion but not yet steel exposure. Red-brown rust indicates zinc coating depletion and active steel corrosion requiring immediate intervention. Localized coating damage from mechanical abrasion, vegetation management equipment contact, or livestock rubbing warrants touch-up treatment before surrounding zinc coating depletes through sacrificial protection of the exposed area.
Vegetation and Debris Management
Maintain minimum 12-inch cleared zone along fence lines to prevent prolonged vegetation contact that traps moisture against wire surfaces. Decomposing plant material generates organic acids that accelerate zinc dissolution. Remove debris accumulation—particularly at fence bases where moisture collection occurs. Avoid herbicide application directly on galvanized wire; drift and overspray of certain formulations (particularly those containing chlorides or strong acids) can damage zinc coatings. Control woody vegetation growth to prevent limb rubbing that abrades coating during wind events. This vegetation management doubles as wildfire fuel reduction in fire-prone regions.
Field Touch-Up Coating Application
Address localized coating damage through field application of zinc-rich repair coatings. Zinc-rich spray coatings containing 69% or higher elemental zinc content (meeting ASTM A780 specification for repair of galvanized coatings) provide cathodic protection equivalent to intact galvanizing when properly applied. Preparation requires wire brushing to remove loose corrosion products and surface contaminants, followed by immediate coating application to clean dry metal. Apply zinc-rich coating to damaged areas plus 2-inch margin beyond visible damage to ensure adequate sacrificial protection. Two thin coats (allowing 30-minute dry time between applications) provide superior performance compared to single heavy coat. Touch-up treatment of mechanical damage points extends fence section service life by 5-10 years at minimal material cost. Target repair efforts at structurally critical locations—line posts, corner braces, and gate attachment points—where premature failure compromises overall fence integrity.
Hardware and Connection Point Protection
Fence hardware including clips, staples, and tie wires often employ lighter galvanizing than primary fence wire, creating preferential corrosion sites. Inspect hardware attachment points annually, replacing corroded fasteners before failure occurs. Ensure galvanized hardware specifications match or exceed fence wire coating class to prevent galvanic corrosion between dissimilar coating thicknesses. At fence durability & lifespan critical junctions, apply zinc-rich coating to hardware during installation as preventive measure.
Common Failure Modes & Corrosion-Related Issues
Crevice Corrosion at Wire Intersections
Crevice corrosion develops in confined spaces—particularly at woven wire knots, where tight contact between crossing wires creates stagnant microenvironments. Moisture infiltrating these crevices cannot readily evaporate, maintaining continuous electrolyte presence. Simultaneously, oxygen depletion within the crevice creates differential aeration cells that accelerate localized corrosion. The geometry of hinge joint and fixed knot configurations inherently creates these crevice conditions. Field observations indicate that crevice corrosion initiates 3-5 years earlier than general surface corrosion on fence wire, making knot integrity the limiting factor in overall fence service life. Class 40 zinc-aluminum coatings demonstrate superior crevice corrosion resistance compared to pure zinc, justifying specification upgrades for critical installations. Proper wire grade selection & tensile properties should account for crevice corrosion susceptibility in lifecycle projections.

Figure 4: Photographic comparison showing corrosion progression on galvanized steel fence wire after 5, 10, and 20 years under different environmental conditions, illustrating coating degradation patterns and failure modes
Bottom Wire Soil Contact Degradation
The lowest horizontal wire in woven fence designs experiences accelerated corrosion due to direct soil contact, soil moisture wicking, and accumulated vegetation debris. Soil chlorides, sulfates, and organic acids concentrate at this interface, creating localized high-corrosivity conditions. Bottom wire failure often occurs 5-10 years before upper wire sections reach end-of-life, potentially requiring partial fence replacement to restore stock containment. Mitigation strategies include specifying one coating class upgrade for bottom wire (Class 40 bottom wire with Class 3 upper wires), elevating the lowest wire 4-6 inches above grade where terrain permits, and maintaining aggressive vegetation clearance in the ground contact zone. Some installations employ stainless steel or polymer-coated bottom wire while using standard galvanized Class 3 for upper wires, balancing cost against service life extension.
Stress Corrosion and Tension Loss
High-tensile fence designs maintain livestock containment through wire tension rather than wire stiffness. Corrosion-induced wire diameter reduction decreases cross-sectional area and effective tensile strength. Simultaneously, corrosion pitting creates stress concentration points vulnerable to fatigue failure under constant tension loading. Field data indicates that tensile strength degradation becomes significant when zinc coating depletion exceeds 50%, typically occurring at 50-60% of projected coating service life. Consequently, fence tension loss and wire breakage often precede visible rust formation, particularly in high-tensile installations operating near design tension limits. Periodic tension monitoring using tension gauges enables proactive wire replacement before catastrophic failure. Initial fence tensioning should account for this anticipated tension loss, installing wire at 80-85% of maximum design tension to accommodate corrosion-related strength reduction over service life.
Differential Corrosion at Hardware Interfaces
Connections between galvanized fence wire and hardware fabricated from dissimilar metals create galvanic corrosion cells when moisture provides electrolyte continuity. Aluminum clips on galvanized steel wire, stainless steel fasteners, or copper grounding connections all establish electrical potential differences driving accelerated corrosion. The less noble metal (typically zinc-coated steel) functions as anode and corrodes preferentially. This galvanic corrosion mechanism concentrates damage at connection points—often the structurally critical fence elements. Specify hardware materials compatible with fence wire galvanizing: zinc-coated steel hardware for galvanized wire, avoiding aluminum or stainless connections unless electrically isolated through polymer bushings or sleeves. When grounding connections to galvanized fence are required (lightning protection, electric fence systems), employ zinc-coated copper lugs or install dielectric isolators to interrupt galvanic cells.
Post-Weld Coating Damage
Welded repairs, custom gate fabrication, or field modifications to galvanized fence wire destroy zinc coating at and near weld points due to high-temperature zinc volatilization. The bare steel weld area initiates immediate corrosion in absence of protective treatment. Weld-affected zones also experience metallurgical changes that increase corrosion susceptibility compared to base wire. Field welding of galvanized fence wire should be minimized through proper initial installation planning. When welding proves necessary, immediately apply zinc-rich repair coating (ASTM A780) to weld and heat-affected zones, overlapping intact galvanizing by minimum 2 inches. Alternatively, specify mechanical connection systems (bolted, clamped) that avoid coating damage altogether. Recognize that field-repaired welds will not achieve the corrosion resistance of intact factory galvanizing; plan for accelerated replacement of welded sections.
Case Studies: Real Field Performance Examples
Case Study 1: 15-Year Class 3 Performance in Temperate Inland Environment
A 12-acre cattle operation in eastern Oklahoma installed 1,500 linear feet of woven wire perimeter fencing in 2009 using Class 3 galvanized wire (0.80 oz/ft² coating weight) with fixed knot construction. The property is located 180 miles from nearest coastline, experiences average annual precipitation of 38 inches, and has moderately acidic clay-loam soils (pH 6.2). After 15 years of service with minimal maintenance—annual mowing along fence line but no coating touch-up—detailed inspection in 2024 revealed the following: upper and mid-height wires retained intact zinc patina with no red rust visible; knot intersections showed early white rust formation indicating zinc corrosion but no steel exposure; bottom wire within 3 inches of soil contact exhibited 20% zinc coating depletion with isolated red rust spots totaling less than 5% of wire length. The fence maintained structural integrity and livestock containment function, with projected remaining service life of 8-12 years before bottom wire replacement becomes necessary. This performance aligns with Class 3 service life projections for moderate inland environments, demonstrating the adequacy of industry-standard coating for typical ranch applications.
Case Study 2: 22-Year Class 40 Performance in Coastal Environment
A horse farm in coastal South Carolina (0.8 miles from Atlantic Ocean) installed perimeter fencing in 2002 employing Class 40 zinc-aluminum coated wire based on prior experience with rapid Class 3 failure in the salt-air environment. The installation encompasses 2,200 linear feet of woven wire fence with 4-inch spacing. The coastal location experiences persistent high humidity (annual average 74%), salt-laden fog events averaging 40 days per year, and aggressive atmospheric corrosion conditions. After 22 years of service with biannual inspection and touch-up coating of mechanical damage points (approximately 15 repair locations over the service period), 2024 inspection documented: galvanized coating remained intact on 90% of wire surface; the characteristic gray ZnAl patina was well-developed; isolated white rust deposits appeared at some knot intersections but without steel exposure; bottom wire sections showed greatest coating depletion (estimated 60-70% of original thickness consumed) but retained adequate protection without red rust development. The fence continues to provide full livestock containment function, with estimated remaining service life of 8-15 years. Standard Class 3 galvanizing in this environment typically fails within 8-12 years; the Class 40 coating has provided approximately double this performance, validating the material cost premium for high-corrosivity installations.
Case Study 3: Premature Failure Due to Inadequate Maintenance
A 40-acre ranch in central Texas installed 3,800 feet of Class 3 galvanized woven wire fence in 2014 but implemented no vegetation management or inspection protocol. Dense woody brush (primarily cedar) was allowed to grow into and through the fence line. After 9 years (2023), catastrophic fence failure occurred when a 200-foot section collapsed due to wire breakage at multiple points. Forensic examination revealed: persistent vegetation contact had abraded zinc coating through mechanical action and trapped moisture against wire surfaces; concentrated corrosion at vegetation contact points had penetrated through zinc coating to steel substrate; steel wire diameter was reduced by 30-40% at failure locations due to extensive pitting corrosion; knot intersections showed complete zinc depletion with advanced steel corrosion undermining wire connection integrity. The observed service life of 9 years represents less than 40% of the expected 25+ year design life for Class 3 wire in this moderate-corrosivity inland environment. This failure exemplifies the critical role of basic maintenance in achieving projected service life—annual vegetation control and biannual inspection would likely have extended this fence to 20+ years of service through early damage detection and touch-up coating application.
Technical Video Resources
Engineering Explanation: This technical video produced by Galco Steel Ireland (2021) demonstrates the hot-dip galvanization process and explains the sacrificial protection mechanism. The presentation includes a field demonstration using a half-galvanized farm gate exposed to weather, clearly showing how rust is halted at the galvanizing boundary with no sideways corrosion creep. The video illustrates the zinc-iron alloy coating formation, barrier protection function, and cathodic protection principles. Particularly valuable for understanding why minor coating damage does not lead to progressive rust failure—a key distinction between galvanizing and organic coatings. Duration: 2 minutes 14 seconds. Suitable for engineers, procurement specialists, and ranch operators evaluating fence wire specifications. View on YouTube
Field Test Documentation: This 2016 video by Premiala documents accelerated corrosion testing comparing hot-dip galvanized steel against various stainless steel grades through multiple exposure protocols: outdoor overnight dew exposure, 10-hour water immersion, dishwasher hot-dry cycles, and concentrated salt spray. The testing methodology includes deliberate coating damage (sandpaper abrasion) to assess corrosion initiation at compromised areas. Results demonstrate that hot-dip galvanized steel develops visible white corrosion (zinc oxide) within the first overnight exposure test, with progressive coating degradation through subsequent test cycles—consistent with zinc’s sacrificial protection function. The video provides valuable visual reference for recognizing zinc patina formation (white deposits) versus steel rust (red-brown), helping field personnel distinguish normal coating behavior from problematic failures. View on YouTube
Industrial Process Overview: Produced by AZZ Incorporated (2025), this video provides step-by-step documentation of commercial hot-dip galvanizing operations, showing surface preparation (degreasing, pickling, fluxing), immersion in molten zinc at 840°F, and post-coating inspection. The presentation explains how iron in steel reacts with molten zinc to form metallurgically bonded zinc-iron alloy layers, distinguishing this process from applied coatings. Relevant for understanding why hot-dip galvanizing provides superior adhesion and abrasion resistance compared to electroplating or spray coatings. The video demonstrates industrial quality control measures ensuring coating uniformity and thickness compliance with ASTM specifications. Duration: 2 minutes 27 seconds. View on YouTube
Additional installation and maintenance guidance is available through installation & corrosion video resources covering field construction techniques that minimize coating damage during fence installation.
FAQ — Corrosion & Galvanized Fencing
Additional corrosion and durability questions are addressed in the comprehensive fence durability & corrosion FAQs resource.
